#207 - High Pressure Showers, Polyaniline, Floating Clay, Epoxy and more...
Water Water Everywhere...
Hey Readers,
After a few false starts, we’re not sure if we are in spring or late winter, one thing is for sure it will never be the season for this!
That said, let’s get into this week’s major innovations and news from around the world of water.
Innovations Roundup
Nanoplastic removal using Epoxy derivatives
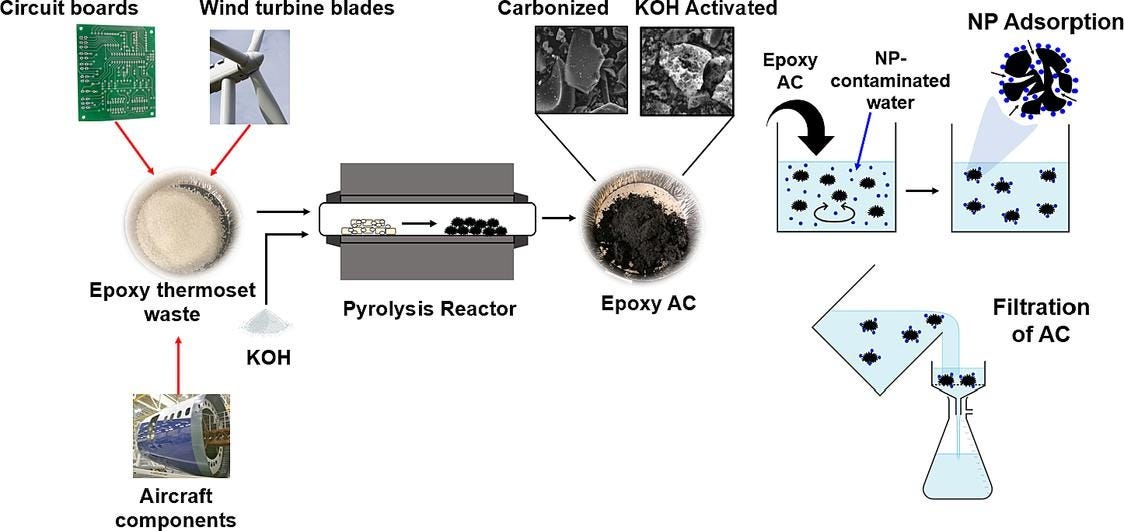
University of Waterloo researchers have created a new technology that can remove harmful nanoplastics from contaminated water with 94% efficiency. Their base product - epoxy, is a waste polymer that can't be reused or reprocessed and often ends up in landfills or finds its way into water system networks like lakes or streams. Using a process called thermal decomposition, the researchers converted epoxy into activated carbon.
They then use the activated carbon to treat water contaminated by nanoplastics. In lab settings they have tested it on nanoparticles of polyethylene terephthalate i.e. PET, most commonly seen in disposable water bottles. The researchers' next steps will look to apply this clean-up process with other types of plastics and scale-up testing in municipal wastewater treatment facilities. Onwards and upwards!
Drug remediation using Floating nanocatalysts on Clay
With more complicated drugs being created to treat a variety of conditions, a good amount of it ends up in our wastewater where it cannot be broken down by traditional water cleaning methods. Additionally the energy required to breakup these drugs lead to folks wanting to harness the power of the sun. Enter a team of Italian researchers who have been looking into ways to make it work with photocatalysts. They first experimented with a powder they added to the contaminated water, but that created something that was only harder to clean up out of the water.
Their solution took the form of a floating catalyst supported by porous beads of clay, which are light enough to stay on top of the water’s surface and thus can easily be removed after use. The clay is chemically inert, meaning it does not react with other materials, and poses no danger to aquatic ecosystems.
For their photocatalyst, the team grew layers of bismuth oxybromide on the beads’ surface. This material is known to have high photocatalytic activity and is chemically stable, minimizing the risk that it would breakdown itself and provide another source of contamination. When in the presence of light, the electrons in the photocatalyst get excited and move towards the outer layer of the material, where they are available to react with carbon-containing molecules to break them down. Simple in sentences, very difficult in practice. They are working on bringing this to a more real world application but nonetheless more power to them!
PFAS detection test - lateral flow testing
A team of chemists from MIT have designed a breakthrough method for the detection of perfluoroalkyl and polyfluoroalkyl substances (PFAS). The experts constructed a sensor capable of identifying these hazardous chemicals in water at remarkably low concentrations (down to 200 parts per trillion in water samples). Their sensor embeds polyaniline, a polymer that transitions between semiconducting and conducting states, onto a nitrocellulose paper strip.
The paper strip is then coated with a surfactant that can pull fluorocarbons such as PFAS out of a drop of water placed on the strip.
When this happens, protons from the PFAS are drawn into the polyaniline and turn it into a conductor, reducing the electrical resistance of the material. This change in resistance, which can be measured precisely using electrodes and sent to an external device such as a smartphone, gives a quantitative measurement of how much PFAS is present. They say this device can be used both by end consumers and in industrial application so it’s a catch-all. Unfortunately the current sensitivity levels of the sensor do not yet meet the stringent EPA guidelines, though the research team is optimistic they will get there.
News Roundup
We have been fans of Source Water for some time now, so it is sad to see them falling down on their commitments to one of their early test communities. It would appear that with their growth and turnover they haven’t been able to support the folks at Allenworth, Arizona. Hope they remedy the situation soon enough.
We’ve seen all the news around using low-flow toilets and smart showers to optimize water but the surprising news from this week was that high pressure showers may actually be better to manage water use. Researchers covertly monitored 290 different showers on the Surrey University campus, and they found that water consumption reduced by up to 60% with higher pressure showers.
And finally for this week, here is a great write-up on the history of Israel’s water efforts, and what are some areas of improvement - laid out by Ali Bin Shahid in his particularly insightful and thorough way. Give his Re:genesis blog a read when you have time.
That is it for this Friday, until next week,
Peace!